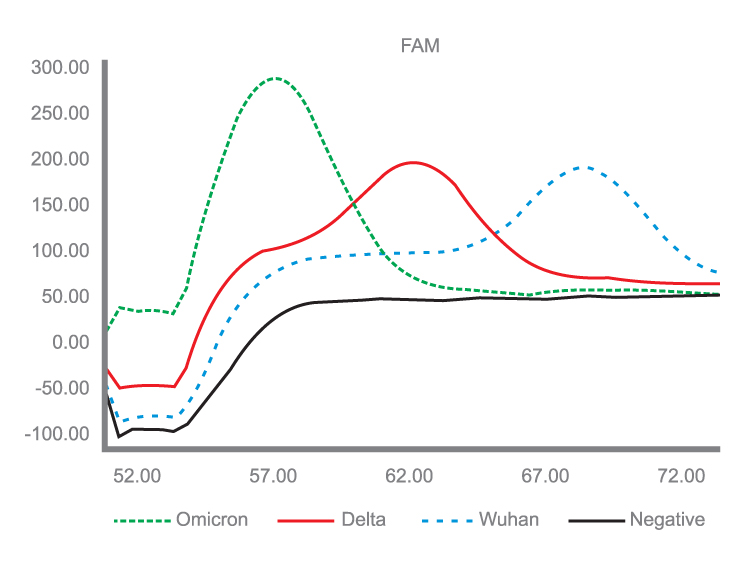
Detection test for the Omicron & Delta Variants of the new COVID-19 Coronavirus (SARS-CoV-2).
Testing for Omicron & Delta Variants of the new Coronavirus is an assay for detecting point mutations in the SARS-CoV-2 spike protein (S).
The test of the presence of Omicron and Delta variants is helpful for identifying therapeutic interventions in symptomatic patients and for the choices of epidemiological measures in asymptomatic carriers of the virus.
The reagent kit used contains special primer and probe systems for the detection of the two SARS-CoV-2 spike protein mutations present in the Delta Variant coronavirus (B.1.617.2). The two detected mutations are L452R and P681R. In addition, the combination of mutations N440K, G446S, S477N, T478K, N679K, and P681H, which are present in the Omicron Variant of SARS-CoV-2 (B.1.1.529) is detected.
The Omicron and Delta Variant test does not distinguish between the BA.1 and BA.2 (stealth version) lineages, which are detected as Omicron variants.
The test is used on samples that have already been analyzed by routine molecular testing (RT-PCR) and found to be positive for coronavirus SARS-CoV-2.
The test is performed with reagents certified for diagnostic use (CE-IVD).